CHAPTER 2
Supplies, equipment, and reagents
Names and descriptions of the reagents, supplies, and equipment necessary for BAC library construction are presented below. Additionally, instructions for making and storing solutions and media are discussed in detail.
Manufacturer/distributor names are given for items/components that (to our knowledge) are (a) produced by a single company, (b) known to be well suited for the application at hand, (c) packaged and sold in quantities/forms that make them especially easy to use, and/or (d) rare or difficult to find. While we have used these products in our research, we do not guarantee their availability, quality, etc., nor do we wish to imply that these products are inherently superior to those of other manufacturers.
Though each of the following chapters contains a list of reagents, supplies, and equipment necessary for the procedures described in that chapter, consult CHAPTER 2 for details regarding these items. Necessary equipment and supplies common to most cell and molecular biology laboratories are not listed at the beginning of individual chapters. However, these items are listed under the subheadings titled "Other" in the sections below.
SUPPLIES
(i.e., disposable items)
- Cheesecloth
- Miracloth
(Calbiochem, cat. no.475855)
- Miracloth squares
: Cut Miracloth into 3 cm2 pieces and autoclave.
- Nitrocellulose filters
(Millipore, cat. no. VSWP 025 00): 0.025 µm pore size
- Plug molds
: Plug molds can be purchased commercially from BioRad. We generally purchase the "disposable" plug molds (cat. no. 170-3713) and re-use them numerous times (see CHAPTER 7 for details). BioRad also sells reusable plug molds (cat. no. 170-3622). However, almost any plastic or glass item into which melted agarose can be poured can serve as a plug mold (e.g., microcentrifuge tubes, plastic pillboxes, pipet tips, small syringes, etc.). If non-standard objects are used as plug molds, cut the resulting plugs into 2 mm x 5 mm x 10 mm rectangles before placing them in lysis buffer.
- Scalpel with #11 blade
- Solution filters
(disposable; Millipore, cat. no. SLGP R25 CS): 0.22 µm pore size; for use in filter sterilization of solutions
- 384-well or 96-well microtiter plates
: These plates can be purchased from several different companies including Nunc, Genetix, and Genome Systems. Because we use a Genetix QBot for clone picking, library replication, gridding, and arraying, we purchase 384-well plates (Genetix, cat. no. X7001) specifically designed for use with a QBot. The Genetix plates have a relatively low profile (which saves valuable freezer space) and are moderately priced (ca. $2.00 per plate).
- Sterile (autoclaved) toothpicks or a hand-held colony picker
: Traditionally, sterile toothpicks have been used in the manual transfer of clones. However, V&P Scientific sells a 12-pin, hand-held colony picker (cat. no. VP 373) that can be used in place of sterile toothpicks. The metal pins of the picker can be flame-sterilized. Use of the hand-held picker reduces the number of movements between X/I/C trays and microtiter plates.
- Library storage boxes
: We store our libraries in cardboard boxes specially designed for holding 36 microtiter plates and/or metal boxes engineered to hold 180 microtiter plates. The cardboard boxes were designed by D. A. Frisch and can be purchased from Southern Container (FIGURE 17.2). The 5-sided metal boxes were designed by A. H. Paterson and can be manufactured by any appropriate metal shop (FIGURE 17.3). The cardboard boxes are less expensive than the metal boxes and have the advantage that stacks of plates are secured on all sides. The metal boxes are sturdier, more space-efficient, and easier to access.
- Other
: pipet tips; 15 ml culture tubes with caps (sterile); 50 ml polypropylene centrifuge tubes (sterile); Pasteur pipets; Kimwipes; Parafilm; plastic wrap; sterile 0.65 ml and 1.5 ml microcentrifuge tubes; weighing boats/paper; 50 ml syringes (for use with solution filters in filter sterilization); autoclavable hazardous waste bags; autoclave tape; aluminum foil; marker pens with ink insoluble in water but soluble in ethanol
EQUIPMENT
Standard kitchen blender
Light microscope: The microscope should be capable of bright-field and/or phase-contrast illumination and have a total magnification power of at least 200x.
Mortar and pestle
CHEF gel apparatus: Instruments suitable for BAC library construction (and general analysis of high molecular weight DNA) are the BioRad CHEF-DR II System (cat. no. 170-3725), the CHEF-DR III Variable Angle System, (cat. no. 170-3700) and the CHEF MapperTM XA System, (cat. no. 170-3670). All systems are equipped with essentially identical electrophoresis chambers, chiller systems, variable speed pumps, combs, casting stands, etc. The three systems differ with respect to the capabilities of their power/control units. Electrophoresis parameters given in the following chapters apply to all three BioRad models.
Large CHEF gel casting stand (BioRad, cat. no. 170-3704): 21 x 14 cm
45-tooth gel comb (for use with the large CHEF gel casting stand): BioRad, cat. no. 170-3645.
30-tooth gel comb (for use with the large CHEF gel casting stand): BioRad, cat. no. 170-3628.
Regular CHEF gel casting stand (BioRad, cat. no. 170-3689): 14 x 13 cm
15-tooth gel comb (for use with the regular CHEF gel casting stand): BioRad, cat. no.170-4324.
UV light box equipped with camera or image capture system: For use in examination and photo-documentation of ethidium bromide-stained agarose gels.
Note 2.1: Always wear appropriate eye and face protection when using a UV light box!
Electroporation system: Electroporation devices are available from several biotechnology companies. See CHAPTER 5 for a discussion of electroporation and APPENDIX A for specific information regarding use of the Gibco BRL Cell-Porator® Electroporation System (cat. no. 11609-013).
Plating rod: A plating rod is a glass bar or tube bent in such a way that it is well suited for spreading liquid across the surfaces of agar plates (see FIGURE 5.1 and FIGURE 5.2).
Plating trays: Square, sterile plating trays (500 cm2) can be purchased from Genetix (Q-Trays, cat. no. X6021). The trays are re-useable. Genetix trays are specifically designed for use with a QBot (a robot designed to pick clones, make BAC grids, etc.). However, these trays also are useful for creating "hand-picked" libraries. If trays are being re-used they must be sterilized prior to adding media. To sterilize used trays, wash trays thoroughly with soap and water, allow to air-dry, spray with 80% ethanol, and place the trays in a single layer in a laminar-flow hood equipped with a UV light for sterilization. Remove the lids from the trays, and stack the lids in the corner of the hood. UV-sterilize the trays for at least 30 minutes. Replace the lids and stack plates on top of each other.
Multi-channel repeat pipettor or automated microtiter plate filler: Such devices are necessary for filling microtiter plates with media. Multi-channel repeat pipettors suited for filling 96-well plates are available from a variety of biotech companies. To our knowledge, there are no multi-channel repeat pipettors for filling 384-well plates. Automated microtiter plate media dispensers allow rapid filling of both 96-well and 384-well plates. They save time and decrease potential contamination problems associated with hand-held pipettors. The Genetix QFill2 (cat. no. QF21) media dispenser can fill a 384-well plate in less than 20 seconds, has an autoclavable manifold and tubing for rapid and thorough clean up, and can be programmed to accurately dispense volumes between 20 µl and 2.0 ml (see APPENDIX C). BioRobotics and Bioneer also sell a comparable automated media dispenser (the PlateFill). V&P Scientific sells a semi-automated plate filling device (the Multi-Spense).
Hand-held plate replicator (optional): Plastic and/or stainless-steel plate replicators are sold by several companies including V&P Scientific, Nunc, Genetix, and Genome Systems.
Electroelution system (optional): See CHAPTER 13 for a discussion of electroelution and APPENDIX B for specific information regarding use of the BioRad Model 422 Electro-Eluter, (cat. no. 165-2976).
Automated DNA isolation system (optional): BAC minipreps can be prepared using standard plasmid isolation techniques (e.g., CHAPTER 6) and/or a number of commercially available kits. In addition, AutoGen sells robots capable of performing BAC isolations from liquid cultures. The Clemson University Genomics Institute (CUGI) uses an AutoGen 740 for performing BAC minipreps.
High-throughput genomics robot (optional): Robots designed specifically for large-scale genomics are available from several companies including Genetix, BioRobotics, GeneMachines, Genomic Solutions, Bioneer, Parallel Biotechnologies, and AutoGen.
The Genetix "QBot" is specifically designed for high-throughput research and is utilized by the Clemson University Genomics Institute (CUGI) (Rod A. Wing, Director) and the University of Georgia AGTEC Center (AGTEC = Applied Genetic Technology) (A. H. Paterson, Director) as well as many other genomic laboratories around the world. The QBot is designed for high speed, high accuracy colony picking, macroarraying, gridding, replicating, rearraying and microarraying. Its picking head, which consists of 96 separately indexable pins, allows picking rates in excess of 3,500 clones per hour. It has automated plate handling, lid lifting, bar code reading, and pin sterilization.
High-throughput genomics robots are not yet standard features in all genomics labs. However, there are several companies and non-profit organizations that offer colony picking, library replication, and library gridding services. These include Genomic Solutions, GeneScreen, AGRF, the Clemson University Genomics Institute (CUGI), the University of Georgia AGTEC Center, Genome Systems, and Bioneer.
- Other
: laminar-flow hood with UV sterilization lamps and Bunsen burner; water baths (16ºC, 37ºC, 45ºC, 65ºC, 70ºC); incubator shaker (37ºC); incubators (30ºC, 37ºC, 50ºC); assorted digital pipettors; stir/hot plates; pH meter; balances; vortex; microwave oven; small submarine gel apparatus (mini-gel apparatus) with power supply; refrigerated centrifuge(s) with rotors/adapters for 500 ml plastic bottles, 50 ml polypropylene centrifuge tubes (conical), and 15 ml culture tubes; microcentrifuge; refrigerator; -80ºC and -20ºC freezers; 4 L plastic beakers; assorted glassware; stir bars; spatulas; autoclave; sources of distilled and molecular biology-grade water; scissors; a large glass or plastic funnel; water bottles
REAGENTS
– Unless otherwise noted, solutions are aqueous.
- MBG (molecular biology-grade) water
: MBG water is sterile water that is essentially free of salts, particulates, and dissolved minerals and metals. It may include "double-distilled" (ddH20) water, deionized water, filtered distilled water, or water purified by other means.
Note 2.2: Unless otherwise noted, all aqueous solutions should be made using MBG water.
- Distilled water
: Distilled water is of intermediate quality between tap water and MBG water.
- 20% SDS (sodium dodecyl sulfate)
: Do not autoclave. Store at room temperature.
- 5 N NaOH stock solution
- Qiagen Large-Construct Kit
(Qiagen, cat. no. 12462): This kit is specifically designed for isolating highly pure BAC DNA.
- Agarose
: Molecular biology grade
- HindIII with 10X buffer and 100X BSA
(New England BioLabs, cat. no. 104L): Enzyme concentration is 20,000 units/ml. The 10X buffer and 100X BSA solutions are supplied with the enzyme.
- HK phosphatase with 10X phosphatase buffer and 0.1 M CaCl2
(Epicentre Technologies, cat. no. H92050): The enzyme concentration is 1.0 units/µl.
- T4 ligase with 10X ligase buffer
(Promega, cat. no. M1804): The enzyme concentration is 3.0 units/µl.
- Blue juice (500 ml)
: Mix 350 ml glycerol, 25 ml 10X TBE, 20 ml 0.5 M EDTA (pH 8.0), 5 ml 20% SDS, and 500 mg bromophenol blue. Add MBG water to 500 ml. Store in aliquots at –20ºC.
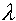 H3 DNA
H3 DNA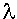 H3 DNA is lambda phage DNA digested to completion with HindIII. It is supplied at a concentration of 500 ng/µl in a buffer consisting of 10 mM Tris-HCl (pH 7.4), 5 mM NaCl, and 0.1 mM EDTA.
H3 DNA is lambda phage DNA digested to completion with HindIII. It is supplied at a concentration of 500 ng/µl in a buffer consisting of 10 mM Tris-HCl (pH 7.4), 5 mM NaCl, and 0.1 mM EDTA. 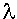 H3 DNA is used as both a size standard and in test ligations.
H3 DNA is used as both a size standard and in test ligations.
- Ethidium bromide
: 10 mg/ml. Store in a light-proof container at room temperature.
Note 2.3: Ethidium bromide should be handled with great care. It is a mutagen/carcinogen!
- 5X TBE
: 0.45 M Tris base (THAM), 2.0% v/v 0.5 M EDTA (pH 8.0) stock solution, 0.44 M boric acid. To make 1.0 L, place 54 g of Tris base, 20 ml of 0.5 M EDTA (pH 8.0), and 27.5 g boric acid in a 1 L bottle. Add MBG water to 1.0 L. Adjust the pH to 8.3.
- 0.5X TBE
: 5X TBE diluted 1:10 with MBG water
- PFGE Lambda Ladder
(New England BioLabs, cat. no. 340): The ladder DNA is embedded in agarose loaded in a 1.0 cc syringe. This ladder is suitable for most PFGE applications.
- PFGE Midrange ladders I and II
(New England BioLabs, cat. no. 355-1 and 355-2, respectively): Because they have more bands than the PFGE Lambda Ladder, the Midrange ladders are of particular use in accurately determining insert sizes from CHEF gels (see CHAPTER 14).
- pBeloBAC11 vector in DH10B (E. coli)
: To obtain pBeloBAC11, write to Dr. H. Shizuya and Dr. M. Simon, Department of Biology, California Institute of Technology, Pasadena, CA 91125, FAX: 808-796-7066 or visit the Caltech Genome Research Laboratory website. Recently BAC vectors derived from pBeloBAC11 have become commercially available (e.g., True-Blue® BAC from Genomics One).
Note 2.4: To prevent confusion, pBeloBAC11 (the most popular BAC vector) will be the only vector discussed in this guide. However, other BAC vectors can be used for cloning, and some vectors are designed for highly specific applications (see Choi and Wing 1999).
- T4 ligase with 10X buffer
(New England BioLabs, cat. no. 202L): Enzyme concentration is 3.0 units/µl.
- CM (chloramphenicol) stock solution
: 5% w/v dissolved in 100% ethanol. To make 20 ml of CM stock solution, dissolve 1.0 g of chloramphenicol in 20 ml of 100% ethanol. Store in a sterile 50 ml polypropylene centrifuge tube at –20ºC.
- Mg2+ stock solution
: 1.0 M MgCl2•6H20, 1.0 M MgS04•7H20. Filter-sterilize and store in sterile container(s) at room temperature.
- 2.0 M glucose stock solution
: Filter-sterilize and store in a sterile container(s) at room temperature.
- 1.0 M KCl stock solution
: Filter-sterilize and store in a sterile container(s) at room temperature.
- SOC (1.0 L)
: Mix 25 g of granulated LB broth (EM Science, cat. no. 1.10285.5000), 10 ml 1.0 M KCl stock solution, and 980 ml MBG water. Adjust pH to 7.0. and autoclave. Cool to room temperature. Immediately before use add 10 ml of Mg2+ stock solution and 10 ml of 2.0 M glucose stock solution.
- LB broth (1.0 L)
: Place 25 g of granulated LB broth (EM Science, cat. no. 1.10285.5000) in a 2.0 L autoclavable bottle. Add MBG water to 1.0 L and mix thoroughly. Adjust solution to pH 7.0 and autoclave. Store at room temperature.
- LB+CM
: Immediately before use, add 250 µl of CM stock solution to 1 L of LB broth (room temperature) to produce LB+CM.
- X-GAL (5-bromo-4-chloro-3-indolyl-beta-D-galactoside) stock solution
: 2% w/v X-GAL dissolved in NN-dimethylformamide. To make 50 ml of X-GAL stock solution, dissolve 1.0 g of X-GAL in 50 ml NN-dimethylformamide. Store in a light proof container at –20ºC.
- IPTG (isopropylthiogalactoside) stock solution
: 20% w/v. To make 10 ml of IPTG stock solution, dissolve 2.0 g of IPTG in 10 ml of MBG water. Filter-sterilize and store in 1.0 ml aliquots at -20ºC.
- X/I/C (X-GAL, IPTG, CM) agar
: 2.5% w/v granulated LB broth (EM Science, cat. no. 1.10285.5000), 1.5% w/v bacto-agar, 0.45% v/v X-GAL stock solution, 0.045% v/v IPTG stock solution, 0.025% v/v CM stock solution. To make 1.0 L of X/I/C agar, place 25 g of granulated LB broth, 15 g of bacto-agar, and a stir bar in a 2.0 L flask. Add MBG water to 1 L and stir until the LB has gone into solution. Cover the top of the flask with aluminum foil and autoclave. Place the mixture on a stir plate and engage the stir bar. Allow the solution to cool to about 50ºC. Transfer the flask to a laminar-flow hood. Gently lift one corner of the foil and add 4.5 ml X-GAL stock solution, 450 µl of IPTG stock solution, and 250 µl of CM stock solution. Swirl the contents of the flask to facilitate mixing. Use the media to prepare X/I/C Petri plates or X/I/C trays as described below.
- X/I/C trays
: Obtain sterile plating trays. If trays are being re-used, wash trays thoroughly with soap and water, dry, and place trays in a single layer in a laminar-flow hood equipped with UV lamps. Remove the lids from the trays and stack the lids in the corner of the hood. UV-sterilize the trays for at least 30 minutes. Replace lids and stack plates. See FIGURE 2.1 for instructions on how to make X/I/C trays using X/I/C agar.
- X/I/C Petri plates
: Set up a laminar-flow hood as described in FIGURE 2.1. Remove the lid from a sterile 90 mm Petri plate. Pour approximately 50 ml of 50ºC X/I/C media into the bottom of the plate. Immediately replace the lid and set the plate in the corner of the hood. Repeat this process until all of the X/I/C media has been poured into plates. Allow the plates to sit in the hood for 2 hours. Carefully wipe any condensation from the inside of each Petri plate lid with a Kimwipe. Invert and stack the plates. The plates can be used immediately, wrapped in aluminum foil and stored (upside down) at room temperature for several days, or wrapped in foil and stored (upside down) at 4ºC for several weeks.
- ElectroMax® DH10BTM competent cells
(Gibco BRL, cat. no. 18290-015): Store at –80ºC. DO NOT ALLOW CELLS TO THAW!
- MP-1 (Miniprep solution 1)
: 50 mM glucose, 25 mM Tris HCl (pH 8.0), 10 mM EDTA. Autoclave and store at 4ºC.
- MP-2 (Miniprep solution 2)
: 0.2 N NaOH, 1.0% w/v SDS. Make immediately before use from 5N NaOH stock solution and 20% SDS stock solution.
- MP-3 (Miniprep solution 3)
: 3.0 M potassium acetate, 28.5% v/v acetic acid. Make immediately before use.
- Isopropanol
: Store at –20ºC
- NotI with 10X buffer and 100X BSA
(New England BioLabs, cat. no. 189L): The enzyme concentration is 10 units/µl.
- MPDB (2-methyl-2,4-pentanediol buffer)
(made day of experiment): 0.5 M 2-methyl-2,4-pentanediol (Aldrich, cat. no. 11,210-0), 10 mM PIPES-KOH, 10 mM MgCl2, 0.5% Triton X-100, 10 mM sodium metabisulfite, 5 mM ß-mercaptoethanol, pH 7.0. To make 50 ml of MPD buffer (enough for a typical experiment), place the following into a 100 ml beaker: 2.95 g of 2-methyl-2,4-pentanediol, 0.15 g of PIPES-KOH, 0.10 g of MgCl2, 0.10 g of sodium metabisulfite, 0.25 ml of Triton X-100, and 19.5 µl of ß-mercaptoethanol. Bring the total volume to 50 ml with MBG water. Add NaOH until the final pH is 7.0. Place the solution in the refrigerator.
- MEB (MPD-based Extraction Buffer)
: 1.0 M 2-methyl-2,4-pentanediol (Aldrich, cat. no. 11,210-0), 10 mM PIPES-KOH, 10 mM MgCl2, 2% polyvinylpyrrolidone (PVP) (Sigma, cat. no. PVP-10), 10 mM sodium metabisulfite, 5 mM ß-mercaptoethanol, 0.5% sodium diethyldithiocarbamate (Sigma, cat. no. D-3506; store at -20°C with dessicant), pH 6.0. Prepare MEB on the day of the extraction, and chill the buffer to 4°C before starting the isolation. To make 3 L of MEB (enough for a typical experiment), place the following into a 4 L plastic beaker: 354.54 g of 2-methyl-2,4-pentanediol (Aldrich, cat. no. 11,210-0), 9.07 g of PIPES-KOH, 6.10 g of MgCl2•6H20, 5.70 g of sodium metabisulfite, 15 g of sodium diethydithiocarbamate (Sigma, cat. no. D-3506; store at -20°C with dessicant), 2 L of distilled water, and 1.2 ml of ß-mercaptoethanol. Mix using a stir bar. Add 60 g of PVP (Sigma, cat. no. PVP-10) a little at a time to prevent formation of PVP clumps. Add water or ice until the total volume is slightly less than 3 L. Add highly concentrated HCl to the medium until the pH is 6.0. If the pH falls below 6.0, add 1N NaOH until the pH is 6.0. Place the solution in the refrigerator.
- TKE (Tris, KCl, EDTA)
: 0.1 M Tris base, 1.0 M KCl, 0.1 M EDTA, pH 9.4-9.5. Autoclave and store at 4°C.
- Liquid nitrogen
- SEB (Sucrose-based Extraction Buffer)
: 10% v/v TKE, 500 mM sucrose, 4 mM spermidine, 1 mM spermine tetrahydrochloride, 0.1% w/v ascorbic acid, 2.0% w/v PVP (MW 40,000), and 0.13% w/v sodium diethyldithiocarbamate (Sigma, cat. no. D-3506; store at -20°C with dessicant). To make 1 L, mix 100 ml TKE, 171.2 g of sucrose, 1.0 g spermidine, 0.35 g spermine tetrahydrochloride, 0.45 ml ß -mercaptoethanol, 1.0 g ascorbic acid, 20.0 g PVP-40, and 1.3 g sodium diethyldithiocarbamate. Add MBG water to 1 L. Make SEB up fresh the day that it is going to be used.
- SEB+BME
: 0.2% v/v ß-mercaptoethanol in SEB. Add ß-mercaptoethanol immediately before use.
- SEB+BME/Triton
: 10% v/v Triton X-100 in SEB+BME
- 100X TE (Tris-EDTA) stock buffer
: 1.0 M Tris base (also known as THAM), 0.1 M EDTA. To make 2 L, place 242.28 g of Tris-base and 58.44 g of EDTA (or 74.44 g of EDTA disodium salt: dihydrate) in a 4 L beaker. Add distilled water to 1.5 L. Adjust pH with concentrated HCl to yield a solution that is pH 7.0. Add distilled water to a final volume of 2 L. Place the 100X TE in glass bottles.
- 1X TE slurry (4.0 L)
: Fill a 4-5 L polypropylene beaker to 3.5 L with crushed ice. Add 40 ml of 100X TE stock buffer. Add distilled water to 4 L.
- 1X TE (non-sterile)
: Dilute the 100X TE stock solution 1:100 with distilled water.
- Percoll
(Amersham Pharmacia, cat. no. 17-0891-01): Percoll consists of silica particles (15-30 nm diameter) coated with PVP. Percoll is non-toxic and virtually chemically inert. When spun in a centrifuge, Percoll forms a density gradient within the range of 1.0-1.3 g/ml. Gradients are iso-osmotic throughout.
- Proteinase K
: This enzyme can be purchased from a variety of molecular biology companies. When purchasing Proteinase K, make sure that the enzyme you order is suitable for molecular biology applications (i.e., molecular biology grade).
- 0.5 M EDTA (pH 9.1) stock solution
: Autoclave and store at room temperature.
- 0.5 M EDTA (pH 8.0) stock solution
: Autoclave and store at room temperature.
- 1.0 M Tris-HCl (pH 8.0) stock solution
: Autoclave and store at room temperature.
- Lysis Buffer
: 1% w/v sodium lauryl sarcosine, 0.1 mg/ml proteinase K, 0.1% w/v ascorbic acid, 2.0% w/v PVP-40, and 0.13% w/v sodium diethyldithiocarbamate (Sigma, cat. no. D-3506; store at -20°C with dessicant) dissolved in 0.5 M EDTA (pH 9.1) stock solution
- Diethyl ether
(4°C)
- Methylene blue
(1.0% w/v)
- LMP (low melting point) agarose
(BMA, SeaPlaque GTG® Agarose, cat. no. 50111)
- 10X uncut lambda DNA
(Amersham Pharmacia, cat. no. 27-4118-01): The DNA comes frozen in solution. The DNA concentration of the solution is 500 µg/ml.
- 1X uncut lambda DNA
: 10X uncut lambda DNA diluted 1:10 in sterile 1X TE
- WB-A (Wash Buffer-A)
: 0.1% w/v ascorbic acid, 2.0% w/v PVP-40, and 0.13% w/v sodium diethyldithiocarbamate (Sigma, cat. no. D-3506; store at -20°C with dessicant) dissolved in 0.5 M EDTA (pH 9.1).
- WB-B (Wash Buffer-B)
: 0.1% w/v ascorbic acid, 2.0% w/v PVP-40, and 0.13% w/v sodium diethyldithiocarbamate (Sigma, cat. no. D-3506; store at -20°C with dessicant) dissolved in 0.05 M EDTA (pH 8.0). Make the 0.05 M EDTA (pH 8.0) by diluting the 0.5 M EDTA (pH 8.0) stock solution.
- WB-C (Wash Buffer-C)
: 0.1% w/v ascorbic acid, 2.0% w/v PVP-40, and 0.13% w/v sodium diethyldithiocarbamate (Sigma, cat. no. D-3506; store at -20°C with dessicant) dissolved in 10 mM EDTA, 10 mM Tris-HCl (pH 8.0). Make the 10 mM EDTA (pH 8.0), 10 mM Tris-HCl (pH 8.0) solution by mixing and diluting the 0.5 M EDTA (pH 8.0) and 1 M Tris-HCl stock solutions.
- 40 mM spermidine
: Dissolve in MBG water, filter sterilize, and store in aliquots at –20°C.
- 1.0 M DTT (dithiothreitol)
: To make 20 ml of 1.0 M DTT, dissolve 3.09 g of DTT in 20 ml of 0.01 M sodium acetate (pH 5.2). Filter-sterilize, aliquot into 1.0 ml microcentrifuge tubes, and store at –20°C.
- H3M (HindIII modified restriction buffer)
: 10% 10X HindIII buffer (supplied with enzyme), 1.0 mM DTT, 4 mM spermidine, 1.0% v/v 100X BSA (supplied with enzyme)
- 0.1 M PMSF (phenylmethylsulfonyl fluoride)
: Dissolve in 100% ethanol and store at -20°C. To make 1.0 ml of 0.1 M PMSF, dissolve 0.017 g of PMSF in 1.0 ml of 100% ethanol. Once in solution PMSF is only effective for 1-2 weeks.
- 70% (v/v) ethanol and 95% (v/v) ethanol
- 50X TAE (Tris, acetic acid, EDTA)
: 2.0 M Tris base (THAM), 5.71% v/v acetic acid, 10% v/v 0.5 M EDTA (pH 8.0) stock solution. To make 1.0 L of 50X TAE, mix 242.2 g of Tris base, 57.1 ml glacial acetic acid, and 100 ml of 0.5 M EDTA (pH 8.0) stock solution in a one liter container. Add MBG water to 1 liter. Store at room temperature.
- 1X TAE
: Mix 1 part 50X TAE with 49 parts MBG water.
- GELaseTM with 50X buffer
(Epicentre Technologies, cat. no. G31200): Enzyme concentration is 0.2 units/µl.
- 10% (w/v) PEG (polyethylene glycol)
: Use molecular biology-grade PEG.
- 40 mM MgSO4 stock solution
: Filter-sterilize and store at room temperature.
- Freezing media
: 2.5% w/v granulated LB broth (EM Science, cat. no. 1.10285.5000), 13 mM KH2PO4, 36 mM K2HPO4, 1.7 mM sodium citrate, 6.8 mM (NH4)2SO4, and 4.4% v/v glycerol. Autoclave and allow media to cool to < 50ºC. In a laminar-flow hood, add 40 mM MgSO4 stock solution to a final concentration of 0.4 mM (MgSO4 will precipitate if it is autoclaved). Immediately before use, add 250 µl of CM stock solution to each liter of freezing media.
- 12% v/v bleach
Return to CONTENTS
Go on to CHAPTER 3